Cryogenics
All about cryogenic technology
Demaco is a specialist in cryogenic infrastructures. Forty years ago, a huge passion arose for this specialist field within the company, that passion has only grown since then. But how exactly is cryogenic technology? What are cryogenic gases, how are ultra-cold temperatures achieved, which risks are involved, and in which industries are cryogenics mostly used? You can read all this, and much more, on this page.
What is cryogenic technology?
Cryogenic technology comes from the Greek word “Kryos” (κρύο), which means “cold”. It is the field in which materials are produced, stored, transported, and used at ultra-cold temperatures. Extreme cold can induce interesting chemical reactions. For example, substances change from gas to liquid or take on a solid form due to cooling.
A well-known liquid that changes its state when cooled is water. At a temperature of 0 ℃, water changes from a liquid into a solid, known as ice. However, this is not cryogenics. It is only when temperatures of -160 ℃ or lower are reached that we speak of cryogenics. These are temperatures at which gases become liquid; this technique is used in various industries.
The temperature required to liquefy a gas varies from one gas to another. For example, oxygen liquefies at a temperature of minus 183 ℃, while helium requires a temperature of at least minus 269 ℃.
Home / Cryogenics
How are cryogenic temperatures achieved?
To achieve freezing temperatures, sophisticated techniques are required. In most cases, four different methods are used to achieve cryogenic temperatures:
1. Thermal conductivity
Thermal conduction is perhaps the most familiar method. By bringing two products or materials into contact, heat transfers from the hottest product to the coldest product. The same principle applies for cryogenic technology. The extreme cold is transmitted by bringing a gas, a liquid, or a solid into contact with a cryogenic liquid. The gas, liquid, or solid, as a result, also reaches the desired cryogenic temperature.
2. Evaporative Cooling
Atoms or molecules have less energy in liquid form than they have in a gaseous form. During the evaporation of a liquid product, atoms or molecules on the surface receive enough energy from the surrounding liquid to convert to its gaseous state. In contrast, the remaining liquid retains less energy, making it colder. Thus, by inducing an evaporation process, the cooling of a liquid can be achieved.
3. Cooling by rapid expansion
A third method is the use of the Joule-Thompson effect. This involves cooling gases by an abrupt expansion of volume or an equally fast pressure drop. This method is extensively used in liquefying hydrogen and helium.
4. Adiabatic demagnetization
The fourth and final method is the use of paramagnetic salts to absorb heat. This method is mainly used within cryogenic technology for cooling liquid helium. Paramagnetic salt can be thought of as a massive number of small magnets that, when placed on a strong magnetic field and processed with an electromagnet, generate or use energy. By absorbing the energy with these materials from a gas, the gas becomes colder and colder.

The history of cryogenic technology
More and more liquid gases emerge
When Demaco was first introduced to cryogenics around 1985, it was a relatively new area of expertise. However, it was not until the 19th century that cryogenics became more widely known, as by then, more and more gases could be successfully liquefied.
It all began back in 1877 when Cailletet and Pictet succeeded in liquefying oxygen. Experiments were in full swing around that time, and soon liquid versions of other gases appeared. For example, in 1884, hydrogen was the first gas to be converted into a mist. In 1892, Sir James Dewar developed a vacuum insulated vessel to store cryogenic liquids, making it easier to work with liquefied gases.
In the following years, experts succeeded in liquefying an increasing number of gases, including the last in line, helium. The liquid form of this gas was first used in 1908.
Cryogenic techniques across a variety of industries
Meanwhile, more and more industries discovered the usefulness of cryogenic technology. In 1961, for example, cryosurgery was first practiced in the United States. Scientists found that slow cooling could destroy unhealthy human tissue. In the United States, liquid nitrogen was used for this purpose, and a few years later, physicians in South Africa also used the method there. In South Africa, however, nitrous oxide was used instead of liquid nitrogen.
The space flight industry also introduced cryogenic technology in the 20th century. In 1961, The American Atlas-Centaur rocket used liquid hydrogen and liquid nitrogen for the first time in the space program. This event is considered a significant milestone in cryogenics and immediately led to large-scale liquid hydrogen production for similar projects.
The medical and aerospace industries are just examples of sectors where cryogenic technology has been in use for a long time already. Cryogenics has also for a long time taken a prominent role in scientific research, the marine industry, and the mass production of liquefied gases in air separation units.
Read on for more on the industries in which cryogenic technologies are used.

Industrial gases
For practical application of cryogenic technology, mainly industrial gases are used.
The leading industrial gases are nitrogen, oxygen, carbon dioxide (CO2), argon, hydrogen, helium, and acetylene. Some of these gases are sold in stores and are available for use by the general public (think helium for balloons and oxygen in the medical field). However, in most cases, cryogenic gases are used by industrial companies.
Cryogenic engineering recognizes several types of industrial gases (some of which are listed in several categories):
Air gases
Air gases are extracted from the air by splitting up different components. The atmosphere consists mainly of nitrogen, oxygen, argon, and a small number of noble gases. Liquid nitrogen is widely used for refrigeration in, for example, the food industry and the medical profession; liquid oxygen is often used in the aerospace industry; liquid argon is best known for its use as a filler gas in light bulbs.
Noble gases
Noble gases (helium, neon, argon, krypton, xenon, and radon) are also found in minimal amounts in the atmosphere. These gases have one thing in common: they hardly react with other materials. Thus, they are often used for cryogenic applications where a reaction (such as toxicity or oxidation) is not desired. For instance, in light bulbs and lasers, and also in balloons and air tanks for divers.
Hydrogen
Hydrogen is also regularly used in cryogenic technology. Hydrogen is the element that, when fused with oxygen, forms water. This gas does not occur on earth in its pure form, but it is regularly produced for the generation of energy or use as a fuel.
Other gases
The above categories cover not all industrial gases. For example, liquid carbon dioxide (CO2) is used in cryogenics but, in our opinion, constitutes an additional category. This gas is on a regular basis mainly used in the food industry.

The risks of working with cryogenic gases
Working with cryogenic temperatures is not without risk. Careless handling in cryogenic technology involves significant risks, like fire and explosion hazards. There is also the risk of gases losing their liquid form and consequently go to waste. Especially in the case of expensive gases, this entails significant financial consequences.
Examples of known risks in the cryogenic field include:
Injury from extreme cold
The cold vapors and gases released from cryogenic liquids can cause skin injuries. In case of prolonged contact with cryogenic materials, the skin can freeze completely and after thawing, leaving behind an irritating abrasion that appears similar to a severe burn.
Dangerous toxics
Most gases have an adverse health effect when they are highly concentrated. Liquid carbon monoxide, for example, can give off carbon monoxide gas, which can be fatal.
Fire and explosion risks
A relatively large number of gases are flammable and explode on contact with fire. Some flammable gases are hydrogen, methane, liquefied natural gas (LNG), and carbon monoxide.
Explosion by rapid expansion
Not only fire, pressure too can cause liquid gases to explode. Without adequately functioning ventilation or overpressure relief devices on cryogenic storage vessels, enormous pressure can build up in these storage vessels or within a pipeline. This can result in a so-called BLEVE (boiling liquid expanding vapor explosion).
The loss of high-value gases
A final risk while working with cryogenic gases is losing them. For gas to stay liquefied, it must be continuously kept at the right temperature. When it accidentally warms up or deteriorates in the infrastructures, loss in gaseous form can occur.
An example of a very precious gas that is easily wasted is helium. Helium is found underground, among several other natural gases. However, to use it, it must be improved to its purest form. This is a costly and laborious process. Also, helium is such a volatile gas that when released, it even leaves the atmosphere.
To prevent the loss of helium as much as possible, Demaco equips all of its helium projects with closed systems. This added protection can make a big difference in saving gas and, by that, the project’s costs.
Good infrastructures in cryogenic engineering
Clearly, cryogenic engineering is a hugely specialized area of expertise, requiring knowledge and skill at the very highest level. With good infrastructures, working with liquid gas is safe; nevertheless, accidents can happen at any time.
Therefore, it is essential that all piping, systems, and other products are designed, built and mounted by an experienced crew who are certified and meet all the quality and safety requirements set forth for cryogenics.

Cryogenic industries
As mentioned earlier, cryogenic equipment is used in a wide range of industries. Over the past decades, Demaco has delivered infrastructures in ten different sectors, and all these industries have their own focus and specific requirements:
1. Automotive
In the automotive industry, cold shrink technology is prevalent. This advanced technology ensures that individual engine components temporarily reach a freezing temperature and shrink slightly, allowing them during assembly to be pressed into the engine with a light force. As the part warms up again, it expands and will be firmly anchored in the engine.
Liquid gases are used to bring parts rapidly to extreme cold, and cryogenic infrastructures are needed to do this.
2. Electronics
A second industry in which cryogenic technology has a significant role is the electronics industry. It is mainly during the testing of electronic devices that extremely cold liquid gases are applied. Liquid nitrogen is commonly used to test whether the equipment can withstand cold temperatures in everyday use.
3. Food
In the food industry, liquid gases are used for cryogenic cooling and the safe packaging of products. Some of the advantages of cryogenic technology in this industry are the processing speed and the fact that products are preserved for a long time while optimally retaining their shape.
4. LNG
The LNG industry is growing at a rapid pace. LNG (Liquefied Natural Gas) offers some environmental advantages for the shipping and road transport sector and is, for that reason, increasingly used as a fuel. To keep LNG liquid, it must be stored at -162 ℃, which requires advanced cryogenic infrastructures.
5. Marine
LNG is used and stored in the marine industry in several ways. Some ships use the gas as fuel, while others transport it in large storage tanks (bunkers). Storage facilities for LNG can also be found in port areas: which are dubbed LNG-plants.
6. Medical
Liquid nitrogen is especially popular in the medical field. This liquid gas is used to perform specific medical treatments and to preserve complex biological structures (this method, called cryopreservation) is used to preserve blood, cells, organs, DNA, and semen).
7. Pharmaceutical
Liquid nitrogen also plays an essential role in the pharmaceutical industry. This liquid gas is widely used to cool cryogenic storage vessels and for molding medicines into pill form.
8. Scientific research
Cryogenic gases, besides being ultra-cold, can also generate tremendous power. For this reason, these gases are often the subject of physics studies. Helium is one of the coldest and most potent liquid gases; therefore, it is widely used to cool particle accelerators.
9. Space
Partly because of the enormous power that cryogenic gases can generate, they are regularly used within the space industry. For example, cryogenic techniques are used to launch satellites and vehicles; liquid gas is fuel (for a cryo engine). At the same time, cryogenics are frequently part of scientific experiments in the space industry.
10. Hydrogen
Interest in hydrogen is growing significantly. Hydrogen is a versatile, clean, and safe energy carrier that can be used as a “fuel” in a fuel cell or as a feedstock in the industrial sector. When hydrogen is combusted, only (water) vapor is released. This makes hydrogen a unique fuel in terms of sustainability. Hydrogen will play an increasingly prominent role in the marine industry, the industrial sector, and the space industry in the coming years.
11. Air separation
A last, but certainly not least, market is the air separation industry. Air separation takes place in air separation units; in short, this means the purification of air for the bulk production of nitrogen, oxygen, argon, and some noble gases.

Cryogenic products
As made clear in the previous sections, cryogenic technologies are common to a vast array of industries. Each industry has its own requirements, challenges, and infrastructures that need to be in place. From piping to controllers, automatic gas vents, and vacuum pumps, the versatility of cryogenic products is virtually endless. However, we would like to list some of the standard products used in cryogenic engineering:
Transfer lines
The transfer line is perhaps the most important product of all in cryogenic engineering. This ultra-insulated pipe system allows liquid gases to be transported safely, without warming up or spilling.
Transfer lines are available in different types, sizes, and materials. For example, vacuum insulated pipes are frequently and widely used because of their high quality and extreme insulation. These pipes can be miles long and often feature diameters from DN10 to 12″.
Next to fixed vacuum insulated pipes, there are also flexible variants. These pipes are ideal for situations in which frequent connecting and disconnecting is required, filling storage vessels (dewars), or for temporary test installations.
Transfer lines are part of almost every cryogenic infrastructure, connected through special couplings that make the handling of liquid gases even safer.
Controllers and sensors
Special controllers and sensors are used to monitor the pressure and liquid level of cryogenic systems. The level sensor automatically monitors the liquid level in quality-improving systems, while the level controller ensures that any accumulated gases will be discharged.
Degassing products
Even with the very best insulation, very small heat inleak can occur in cryogenic systems. Because of this inleak and pressure drops in the system, some of the gas will evaporate. This often results in gas accumulations, which impedes the flow.
Fortunately, there are some handy products available that efficiently discharge these gases. For example, by connecting an automatic degasser vent to the highest point of a nitrogen system, the accumulated gas will be vented out of the system at the time of no or low-flow.
To prevent ice buildup at the end of a gas line, a degasser heater can be installed. The heater only heats the out-flow section of the pipe.
Phase separators
To ensure that liquid gases remain as pure as possible and gas-forming can be discharged, phase separators are installed. By using an internal storage vessel, this quality-improving product ensures that liquid gas is temporarily halted. Since any gas is lighter than its liquid form, any gas bubbles at a standstill (or at a minimal flow rate) will naturally rise upwards, allowing them to be easily discharged.
The use of a phase separator improves the quality of liquid gases, ensures a sufficient supply of liquid gas, and allows the pressure to be lower than the storage tank’s pressure.
Subcoolers
Another commonly used quality improvement product is the subcooler. The Demaco subcooler is used to cool liquefied gas so that gas is not formed in the pipes during operation. Any formed gas bubbles are separated from the liquid gas through an advanced heat exchange process, thus ensuring that only high quality pure liquid gas remains.
Cryogenic solutions for specific industries
Next to products that regulate the supply, storage, and quality of liquid gases, various products are available that facilitate the application of these products for specific industries.
Some examples include liquid nitrogen filling stations, cold boxes for shrink-fitting automotive parts, and quality improvement products that allow liquid nitrogen to be used without instant evaporation.
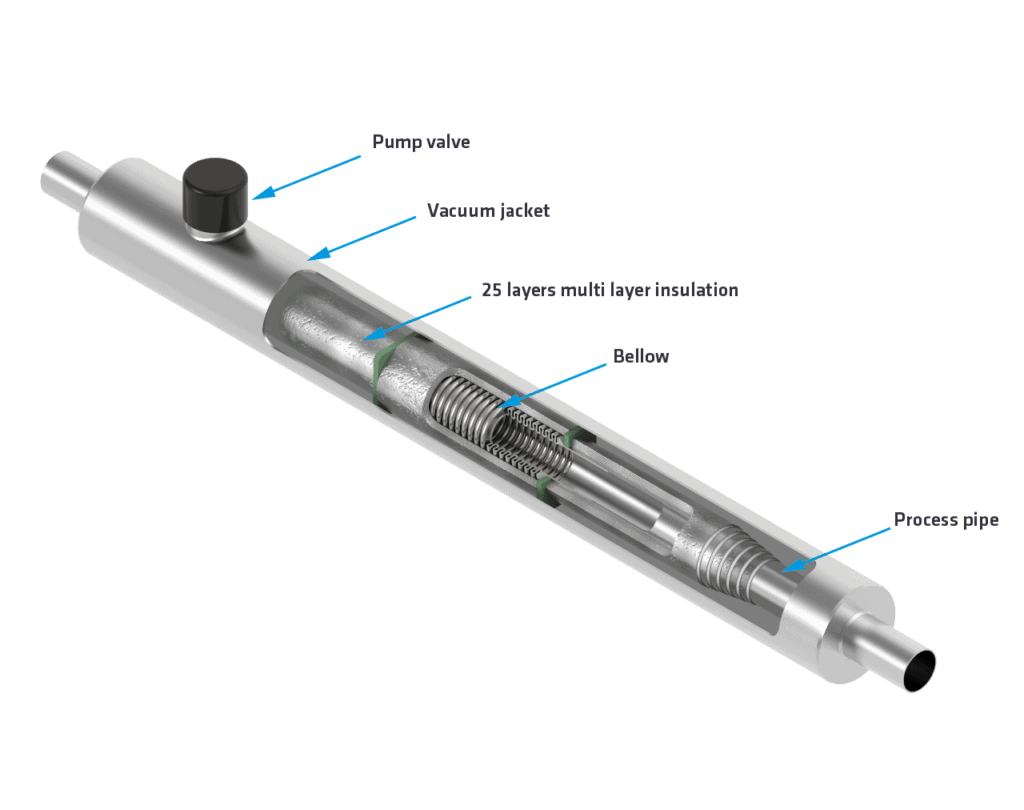
Vacuum technology
Insulation plays an essential role within cryogenic technology. Cryogenic gases must be kept at the perfect temperature for their usability in liquid form, which is not that easy. The transportation of liquid gases sometimes involves several kilometers of travel, moving the gas from, let’s say, a terminal, through various pipelines to a refrigeration system, a ship, a launching pad, or any other destination.
During each step and throughout the entire logistical process, the chances are that the gas will heat up and is lost. A small heat leak can have a significant impact, and this renders optimal cryogenic insulation extremely important.
In recent years, vacuum technology has proven to be the best technique for maintaining the (ultra-cold) temperature of transfer lines and cryogenic systems. Compared to conventional insulation, the insulation value of vacuum insulation is much higher, which means that the heat inleak is less and fewer gases are lost. Compared to insulation made of, for example, PIR/PUR, Foamglas, Armaflex, Perlite, and Misselon, vacuum insulation offers an insulation value that is 15 times higher than any other method.

Demaco is an expert in cryogenic engineering
Our cryogenic engineers for decades have been committed to being the best in designing and building cryogenic infrastructures. As true Cryogenius’, the passionate Demaco team knows all about cryogenic gases and their specific application within various industries.
We advise, design, build, manufacture, supply, assemble, supervise, and among other things, certify piping, systems, and vacuum insulated components.
Please take a look at our products and services pages for more information about our services and products. For everything about vacuum technology and our vacuum insulated products, please visit our Vacuum Webshop.
